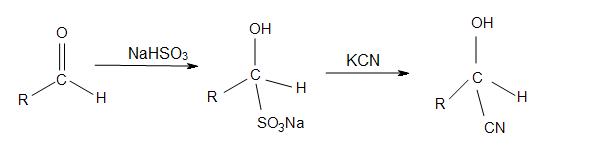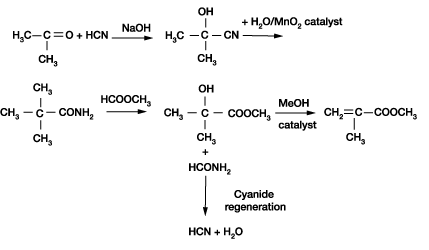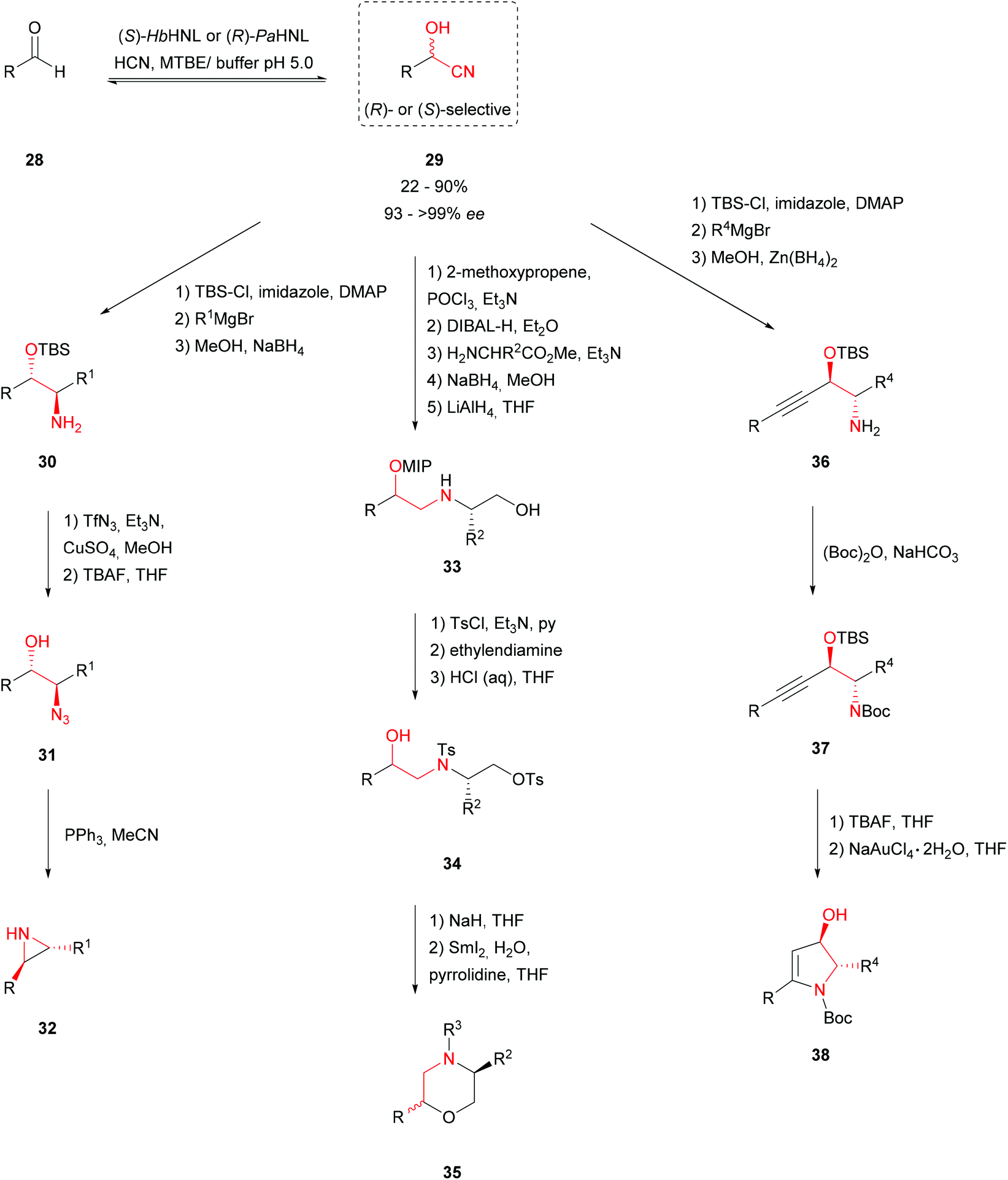
Enantioselective synthesis of cyanohydrins catalysed by hydroxynitrile lyases – a review - Organic & Biomolecular Chemistry (RSC Publishing) DOI:10.1039/C6OB00934D
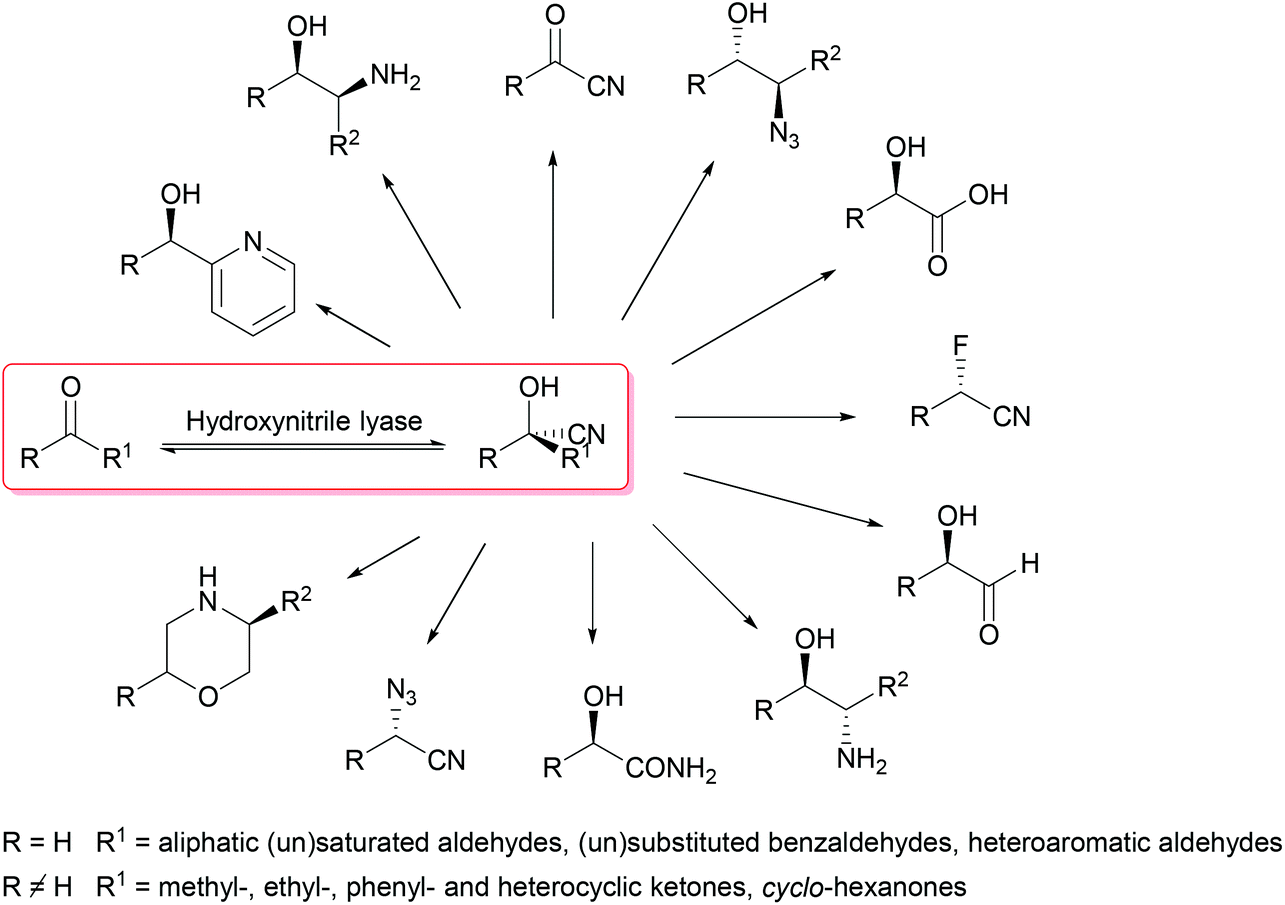
Enantioselective synthesis of cyanohydrins catalysed by hydroxynitrile lyases – a review - Organic & Biomolecular Chemistry (RSC Publishing) DOI:10.1039/C6OB00934D

Enantioselective synthesis of cyanohydrins catalysed by hydroxynitrile lyases – a review - Organic & Biomolecular Chemistry (RSC Publishing) DOI:10.1039/C6OB00934D
_web.jpg)
Short Topic: More ways to use reagents | Mitsunobu Reaction Using Acetone Cyanohydrin | TCI EUROPE N.V.

Acetone Cyanohydrin as a Source of HCN in the Cu-Catalyzed Hydrocyanation of α-Aryl Diazoacetates | The Journal of Organic Chemistry

Catalytic Transfer Hydration of Cyanohydrins to α-Hydroxyamides | Journal of the American Chemical Society

Acetone cyanohydrin - 75-86-5, C4H7NO, density, melting point, boiling point, structural formula, synthesis

Preparation of acetone cyanohydrin (2-hydroxy-2-methylpropanenitrile; 2-hydroxyisobutyronitrile; alpha-hydroxyisobutyronitrile)